|
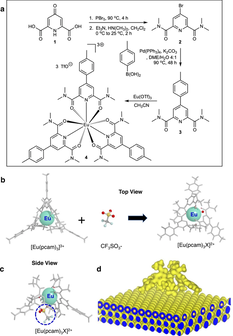
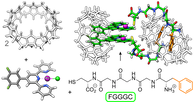
Ajayi, T. M.; Singh, V.; Latt, K. Z.; Sarkar, S.; Cheng, X.; Premarathna, S.; Dandu, N. K.; Wang, S.; Movahedifar, F.; Wieghold, S.; Shirato, N.; Rose, V.; Curtiss, L. A.; Ngo, A. T.; Masson, E.; Hla, S. W. Nat. Commun., 2022, 13, 6305. Atomically precise control of rotational dynamics in charged rare-earth complexes on a metal surface. DOI: 10.1038/s41467-022-33897-3 |
|
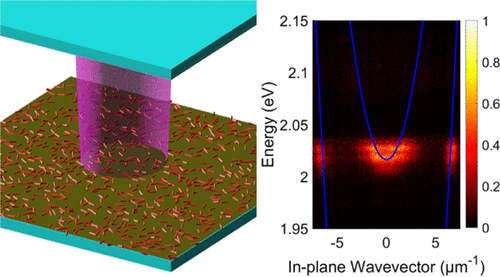
Emmanuele, R.; Maciejczyk, M.; Smith, A.; Cheng, X.; Masson, E.; Gosztola, D. J.; Hla, S. W.; Robertson, N.; Ma, X. ACS Photonics, 2022, 9, 2315–2321.
Microcavity-Modified Emission from Rare-Earth Ion-Based Molecular Complexes. DOI: acsphotonics.2c00289 |
|
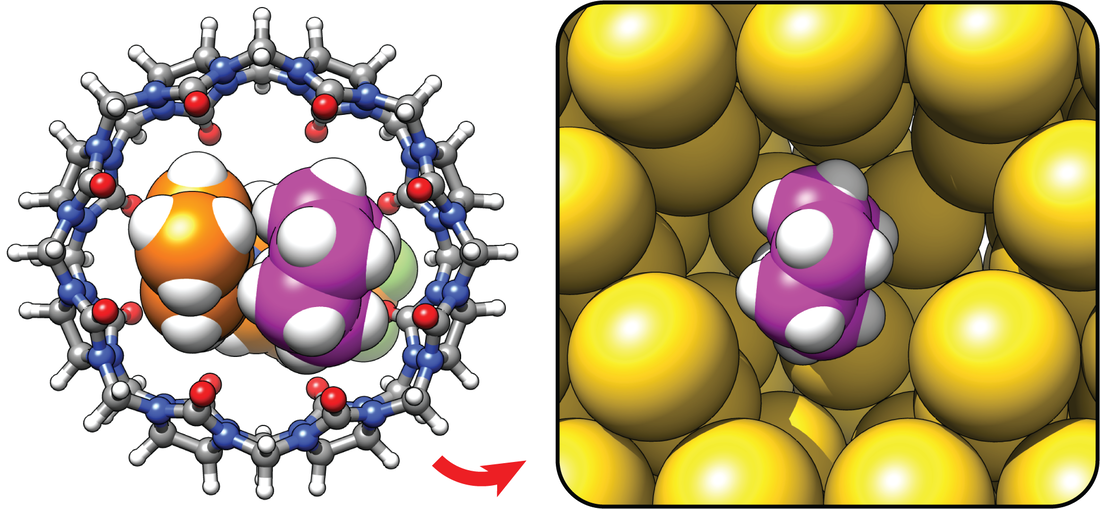
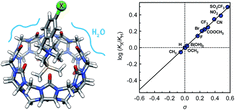
Nazimuddin, Md; Barbero, H.; Rabbani, R.; Masson, E. Chem. Sci., 2022,13, 4388-4396.
Cucurbiturils mimicked by low polarizability solvents with pre-formed cavities: an empirical model to predict hydrocarbon selectivity. DOI: 10.1039/D1SC06728A |
|
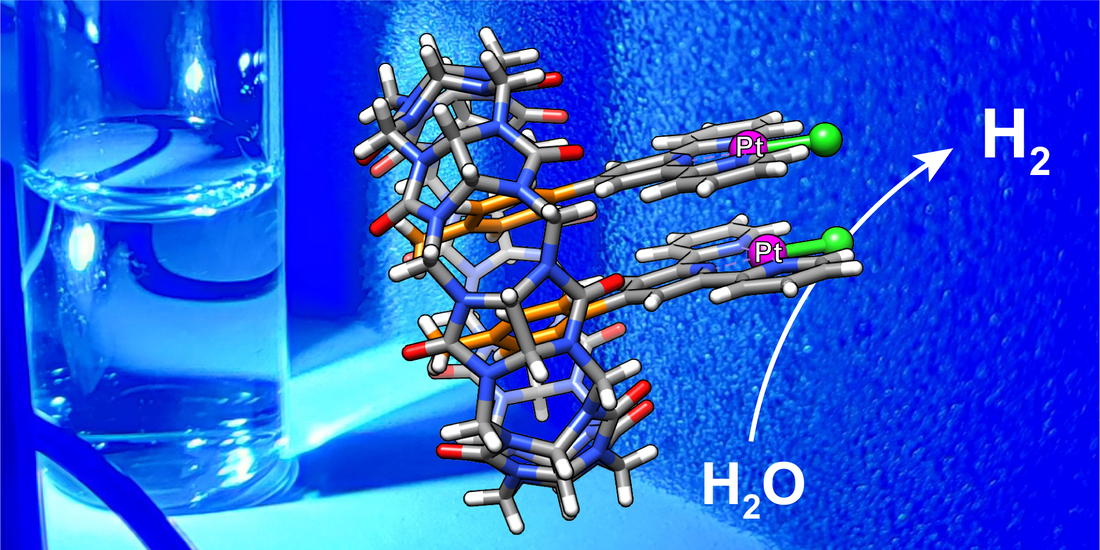
Rabbani, R.; Saeedi, S.; Nazimuddin, Md; Barbero, H.; Kyritsakas, N.; White, T. A.; Masson, E. Chem. Sci., 2021,12, 15347-15352.
Enhanced photoreduction of water catalyzed by a cucurbit[8]uril-secured platinum dimer. DOI: 10.1039/D1SC03743A |
|
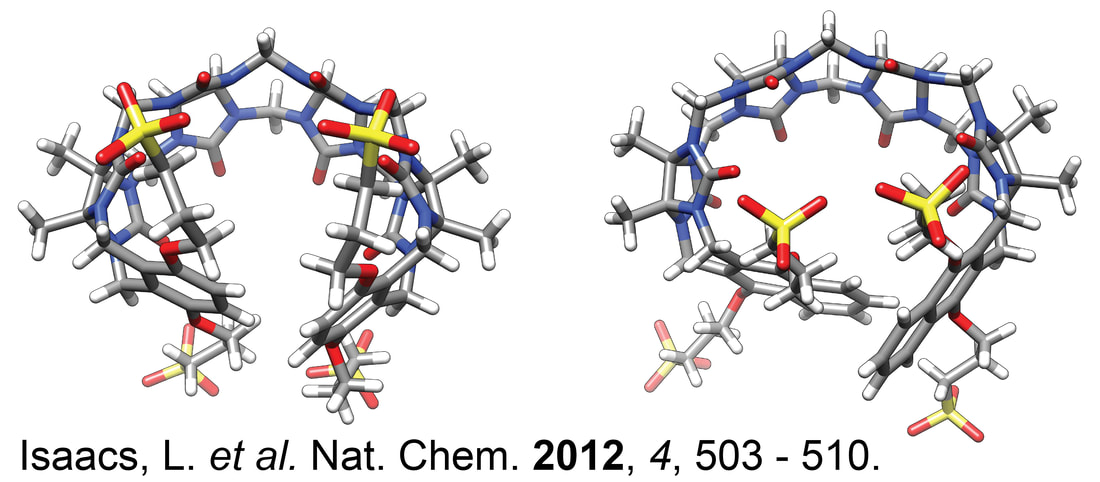
Barbero, H.; Masson, E. Chem. Sci., 2021, 12, 9962-9968.
Design and recognition of cucurbituril-secured platinum-bound oligopeptides. DOI: 10.1039/D1SC02637B |
|
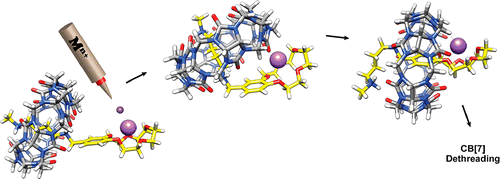
Dračínský, M.; Hurtado, C. S.; Masson, E.; Kaleta, J. Chem. Commun., 2021, 57, 2132-2135.
Stuffed pumpkins: mechanochemical synthesis of host–guest complexes with cucurbit[7]uril. DOI: 10.1039/D1CC00240F |
|
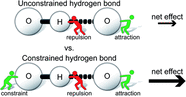
Barbero, H.; Meunier, A.; Kotturi, K; Smith, A.; Kyritsakas, N.; Killmeyer, A.; Rabbani, R.; Nazimuddin, Md; Masson, E. Phys. Chem. Chem. Phys., 2020, 22, 20602-20611.
Counterintuitive torsional barriers controlled by hydrogen bonding. DOI: 10.1039/D0CP03285A |
|
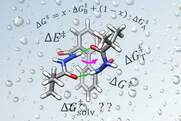
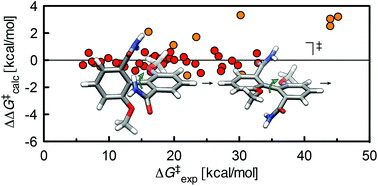
Vik, E. C.; Li, P.; Maier, J. M.; Madukwe, D. O.; Rassolov, V. A.; Pellechia, P. J.; Masson, E.; Shimizu, K. D. Chem. Sci., 2020, 11, 7487-7494.
Large transition state stabilization from a weak hydrogen bond. DOI: 10.1039/D0SC02806A |
|
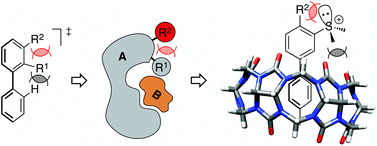
Barbero H.; Thompson, N. A.; Masson E. J. Am. Chem. Soc., 2020, 142, 867 – 873.
“Dual Layer” Self-Sorting with Cucurbiturils. DOI: 10.1021/jacs.9b09751 |
|
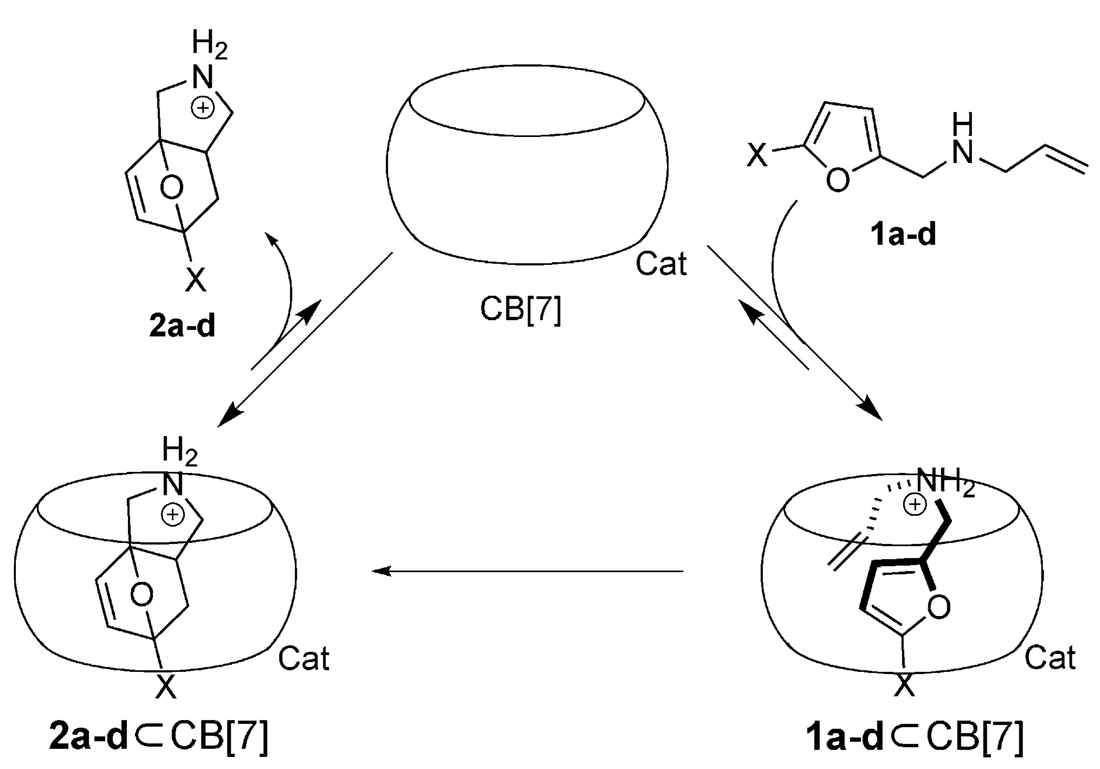
Thompson, N. A.; Barbero H.; Masson E. Chem. Commun., 2019, 55, 12160 – 12163.
Templating conformations with cucurbiturils. DOI: 10.1039/C9CC06766C |
|
Barbero, H.; Masson, E. Monographs in Supramolecular Chemistry No. 28. Cucurbiturils and Related Macrocycles, 2019, Ed. Kim, K.; RSC.
Cucurbiturils as Reaction Vessels. DOI: 10.1039/9781788015967-00086 |
|
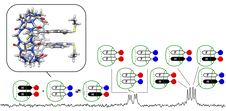
Raeisi, M.; Kotturi, K.; del Valle, I.; Schulz, J.; Dornblut, P.; Masson, E. J. Am. Chem. Soc. 2018, 140, 3371 – 3377.
Sequence-Specific Self-Assembly of Positive and Negative Monomers with Cucurbit[8]uril Linkers. DOI: 10.1021/jacs.7b13304 |
|
Kotturi, K.; Masson, E. Chem. Eur. J. 2018, 24, 8670 – 8678.
Directional Self-Sorting with Cucurbit[8]uril Controlled by Allosteric π-π and Metal-Metal Interactions. DOI: 10.1002/chem.201800856 |
|
Premadasa, U. I.; Moradighadi, N.; Kotturi, K.; Nonkumwong, J.; Khan, Md. R.; Singer, M.; Masson, E.; Cimatu, K. L. A. J. Phys. Chem. C 2018, 122, 16079 – 16085.
Solvent isotopic effect on surfactant headgroup at air-liquid interface. DOI: 10.1021/acs.jpcc.8b03680 |
|
Masson, E.; Raeisi, M.; Kotturi, K. Isr. J. Chem. 2018, 58, 413 – 434.
Kinetics Inside, Outside and Through Cucurbiturils. DOI: 10.1002/ijch.201700120 |
|
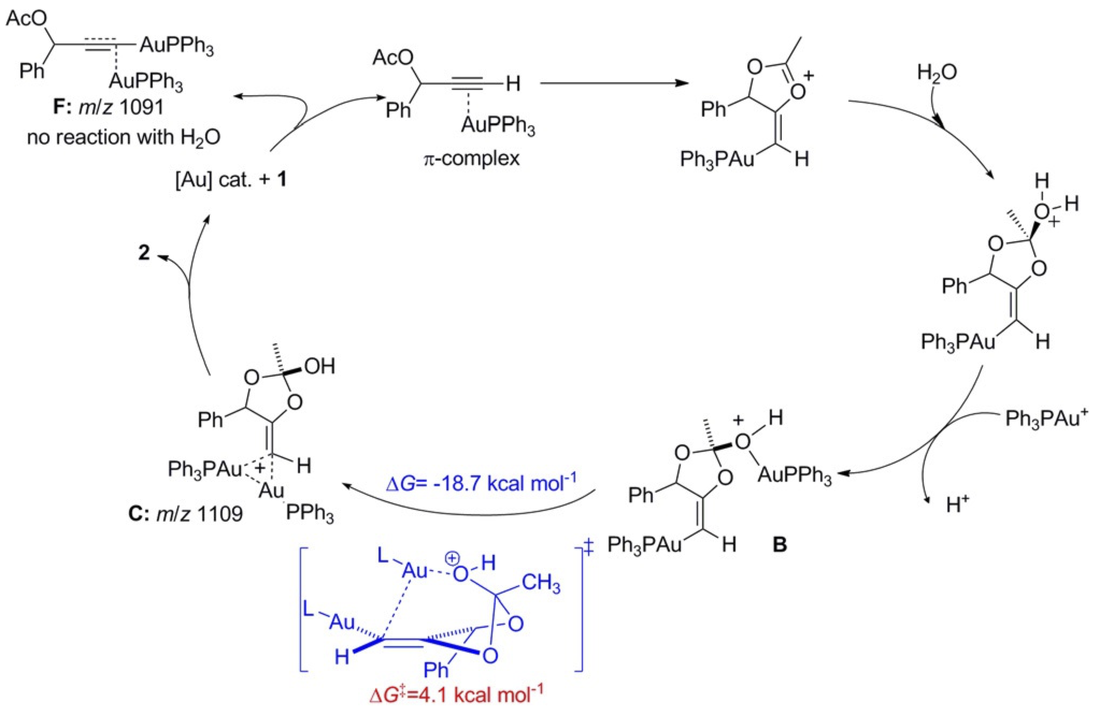
Lu, M.; Su, Y.; Zhao, P.; Ye, X.; Cai, Y.; Shi, X.; Masson, E.; Li, F.; Campbell, J. L.; Chen, H. Chem. Eur. J. 2018, 24, 2144 – 2150.
Direct Evidence for the Origin of Bis-Gold Intermediates: Probing Gold Catalysis with Mass Spectrometry. DOI: 10.1002/chem.201703666 |
|
Palma, A.; Artelsmair, M.; Wu, G.; Lu, X.; Barrow, S. J.; Uddin, N.; Rosta, E.; Masson, E.; Scherman, O. A. Angew. Chem., Int. Ed. 2017, 56, 15688 – 15692.
Cucurbit[7]uril as a Supramolecular Artificial Enzyme for Diels-Alder Reactions. DOI: 10.1002/anie.201706487 |
|
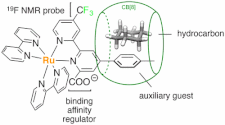
Rabbani, R.; Masson, E. Org. Lett. 2017, 19, 4303 – 4306.
Probing Interactions between Hydrocarbons and Auxiliary Guests inside Cucurbit[8]uril. DOI: 10.1021/acs.orglett.7b01966 |
|
Masson, E., Reference Module in Chemistry, Molecular Sciences and Chemical Engineering, Comprehensive Supramolecular Chemistry II, 2017, 5.03, 21-45. Ed. Atwood, J. L.; Elsevier
Biomedical Applications of Cucurbiturils and Derivatives. DOI: 10.1016/B978-0-12-409547-2.12560-0 |
|
Ling, X.; Saretz S.; Xiao, F.; Francescon, J.; Masson, E. Chem. Sci. 2016, 7, 3569 – 3573.
Water vs. cucurbituril rim: a fierce competition for guest solvation. DOI: 10.1039/C5SC04475H |
|
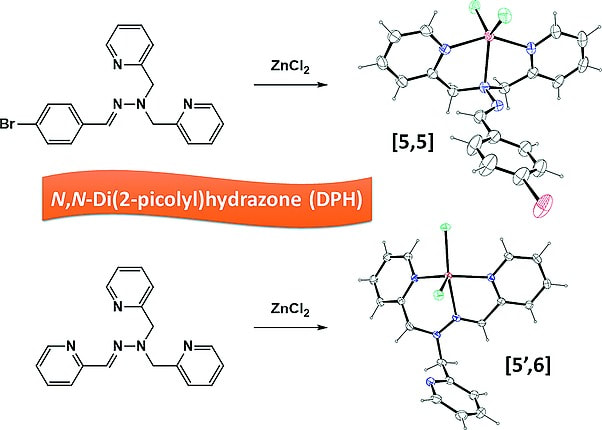
Barsoum, D. N.; Kyeremeh-Mensah, L; Meisner, Q. J.; Clark, R. J.; Masson, E.; Zhu, L. Eur. J. Inorg. Chem. 2016, 35, 1434 – 1448.
Zinc(II) Complexes of N,N-Di(2-picolyl)hydrazones. DOI: 10.1002/ejic.201601066 |
|
Masson, E.; Schlosser, M. (in memoriam) Arkivoc 2015, 139 – 150.
Are reactions between allylpotassiums and halotrimethylsilanes diffusion-controlled? DOI: 10.3998/ark.5550190.p009.019 |
|
Joseph, R.; Nkrumah, A.; Clark, R. J.; Masson, E. J. Am. Chem. Soc. 2014, 136, 6602 – 6607.
Stabilization of Cucurbituril/Guest Assemblies via Long-Range Coulombic and CH···O Interactions. DOI: 10.1021/ja4092165 |
|
Joseph, R.; Masson, E. Supramol. Chem. 2014, 26, 632 – 641.
Cucurbit[8]uril recognition of rapidly interconverting diastereomers. DOI: 10.1080/10610278.2014.934243 |
|
Joseph, R.; Masson, E. Eur. J. Org. Chem. 2014, 105 – 110.
Atropisomerization in Confined Space; Cucurbiturils as Tools to Determine the Torsional Barrier of Substituted Biphenyls. DOI: 10.1002/ejoc.201301460 |
|
Joseph, R.; Masson, E. Org. Biomol. Chem. 2013, 11, 3116 – 3127.
Subtle "supramolecular buttressing effects" in Cucurbit[7]uril/guest assemblies. DOI: 10.1039/C3OB40250A |
|
Masson, E. Org. Biomol. Chem. 2013, 11, 2859 – 2871.
Torsional barriers of substituted biphenyls calculated using density functional theory: a benchmarking study. DOI: 10.1039/C3OB26704K |
|
Masson, E.; Ling, X.; Joseph, R.; Kyeremeh-Mensah, L.; Lu, X. RSC Advances 2012, 2, 1213 – 1247.
Cucurbituril Chemistry: A tale of supramolecular success. DOI: 10.1039/C1RA00768H |
|
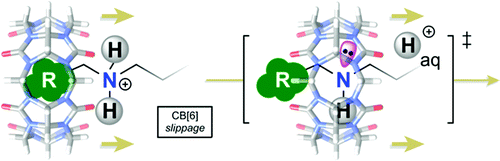
Ling, X.; Masson, E. Org. Lett. 2012, 14, 4866 – 4869.
Cucurbituril slippage: Cations as Supramolecular Lubricants. DOI: 10.1021/ol3021989 |
|
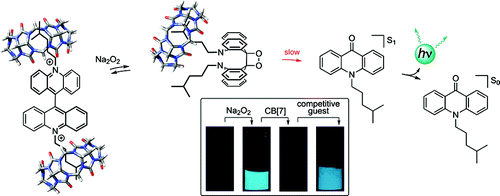
Masson, E.; Shaker, Y. M.; Masson, J. P.; Kordesch, M. E.; Yuwono, C. Org. Lett. 2011, 13, 3872 – 3875.
"Supramolecular Circuitry”: Three Chemiluminescent, Cucurbit[7]uril-Controlled On/Off Switches. DOI: 10.1021/ol201403m |
|
Lu, X.; Masson, E. Langmuir 2011, 27, 3051 – 3058.
Formation and Stabilization of Silver Nanoparticles with Cucurbit[n]urils (n = 5−8) and Cucurbituril-Based Pseudorotaxanes in Aqueous Medium. DOI: 10.1021/la104729j |
|
Ling, X.; Samuel, E. L.; Patchell, D. L.; Masson, E., Org. Lett. 2010, 12, 2730 – 2733.
Cucurbituril Slippage: Translation is a Complex Motion. DOI: 10.1021/ol1008119 |
|
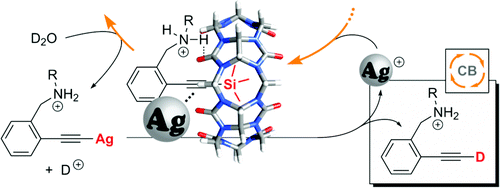
Lu, X.; Masson, E., Org. Lett. 2010, 12, 2310 – 2313.
Silver-Promoted Desilylation Catalyzed by Ortho- and Allosteric Cucurbiturils. DOI: 10.1021/ol100667z |
|
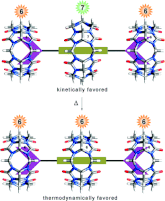
Masson, E.; Lu, X.; Ling, X.; Patchell, D. L. Org. Lett. 2009, 11, 3798 – 3801.
Kinetic vs Thermodynamic Self-Sorting of Cucurbit[6]uril, Cucurbit[7]uril, and a Spermine Derivative. DOI: 10.1021/ol901237p |